Seattle biotech firm is the primary to obtain approval to check B cell gene remedy in people
Seattle biotech startup Immusoft has received approval to begin clinical trials of its novel strategy for treating genetic disease, the company announced Thursday. Immusoft says it’s the first to get permission to use engineered immune system cells called B cells in a human study.
The U.S. Food and Drug Administration (FDA) approved Immusoft’s Investigational New Drug Application for testing its immunotherapy for a rare, lethal childhood disease called MPS I.
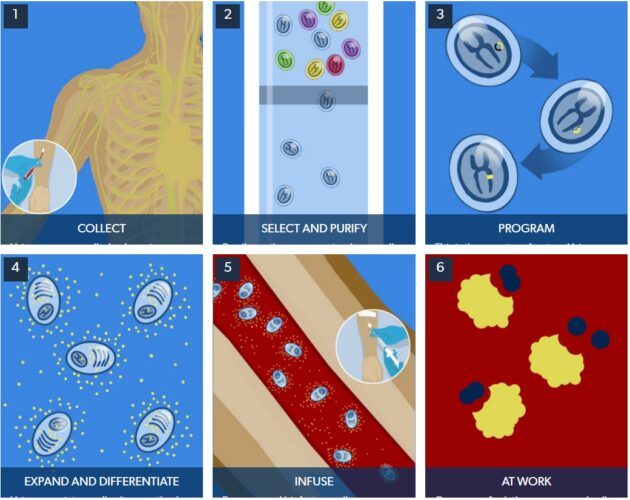
An immune response includes a suite of players, and the B cells are responsible for producing the antibodies that stick to invading bacteria and viruses. The company is able to modify B cells into “biofactories” that instead crank out missing or non-functioning enzymes and proteins in the cells of patients.
“This is a huge achievement for the company and a historic moment in the field of cell and gene therapies,” said Sean Ainsworth, Immusoft’s CEO and chairman, in a statement.

The approach has potential advantages to current strategies for delivering treatments. Therapies that use a virus as its delivery mechanism can trigger immune responses that limit their effectiveness. Treatments using stem cells can have difficulties associated with chemotherapy and stem cell transplants.
Immusoft’s technology, called ISP-001, uses a patient’s own B cells, reprogramming them to make needed proteins. Other companies working on B cell therapies include Be Biopharma and Walking Fish Therapeutics.
“I don’t know if they are going to be successful, but it’s exciting for all of us that they have gotten permission to start a trial,” researcher Richard James told MIT Technology Review. James’s lab at the University of Washington is also working on B cell engineering.
The trial will be done at the University of Minnesota Medical School and led by Dr. Paul Orchard, a professor in the university’s Division of Pediatric Bone Marrow Transplantation.
Children with MPS I are not able to produce an essential enzyme that helps break down long-chain sugars inside cells. The sugars then build up in cells, causing progressive damage. Severe MPS1 occurs in about 1 in 100,000 births, and symptoms appear within a year.
Immusoft is interested in expanding its therapy to other rare diseases, as well as cardiovascular, autoimmune and central nervous system diseases.
In October 2021, the company announced a collaboration with pharma giant Takeda to develop treatments targeted to the nervous system in a deal worth potentially more than $900 million.
Immusoft was founded in 2009 and has raised more than $50 million in venture capital, according to PitchBook. In 2018, Ainsworth took over leadership from founder Matthew Scholz.
Scholz is now CEO of Oisín Biotechnologies, a startup developing preclinical therapies that target and kill damaged “zombie” cells. He is still on Immusoft’s board of directors.
Conclusion: So above is the Seattle biotech firm is the primary to obtain approval to check B cell gene remedy in people article. Hopefully with this article you can help you in life, always follow and read our good articles on the website: Doshared.com